Latest Posts
-

The science behind successful new product development: from concept to market
49 views -

How to get your first electric bike
47 views -
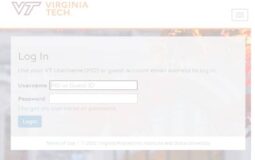
Hokie SPA Login @hokiespa.vt.edu
2,205 views -

Shadow health login
1,736 view